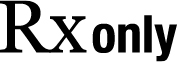| Manufacturer | Indicates the medical device manufacturer | 5.1.1 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Authorized representative in the European Community | Indicates the authorized representative in the European community | 5.1.2 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Catalogue number | Indicates the manufacturer's catalogue number so that the medical device can be identified | 5.1.6 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Sterilized using ethylene oxide | Indicates a medical device that has been sterilized using ethylene oxide | 5.2.3 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Do not resterilize | Indicates a medical device that is not to be resterilized | 5.2.6 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Non-sterile | Indicates a medical device that has not been subjected to a sterilization process | 5.2.7 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
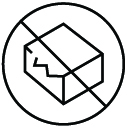 | Do not use if package is damaged | Indicates a medical device that should not be used if the package has been damaged or opened | 5.2.8 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
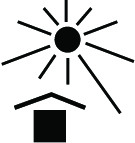 | Keep away from sunlight | Indicates a medical device that needs protection from light sources | 5.3.2 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Keep dry | Indicates a medical device that needs to be protected from moisture | 5.3.4 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Do not re-use | Indicates a medical device that is intended for one use, or for use on a single patient during a single procedure | 5.4.2 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Consult instructions for use | Indicates the need for the user to consult the instructions for use | 5.4.3 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | Caution | Indicates the need for the user to consult the instructions for use for important cautionary information such as warnings and precautions that cannot, for a variety of reasons, be presented on the medical device itself | 5.4.4 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | In vitro diagnostic medical device | Indicates a medical device that is intended to be used as an in vitro diagnostic medical device | 5.5.1 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
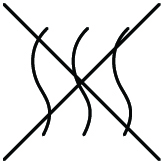 | Non-pyrogenic | Indicates a medical device that is non-pyrogenic | 5.6.3 | ISO 15223-1:2016 Medical Devices - Symbols to be used with medical device labels, labeling and information to be supplied - Part 1: General Requirements |
 | CE registration mark | Indicates that the medical device conforms with the health, safety and enviromental standards for products sold with the European Economic Area (EEA) | N/A | N/A |
 | CE0123 registration mark | Indicates that the medical device is in compliance with the CE Medical Device Directive 93/42/EEC | N/A | N/A |
 | Medical Device | Indicates that the product is classified as a medical device | N/A | N/A |
 | No Latex | Indicates the medical device does not contain the presence of natural rubber or dry natural rubber latex as a material of construction within the medical device or its packaging | N/A | N/A |
 | Prescription only | Caution: U.S. Federal Law restricts this device to sale by or on the order of a physician | N/A | N/A |
 | Carton Quantity | Indications the number of units contained in the carton | N/A | N/A |
 | Carton Weight | Indicates the weight of the carton | N/A | N/A |